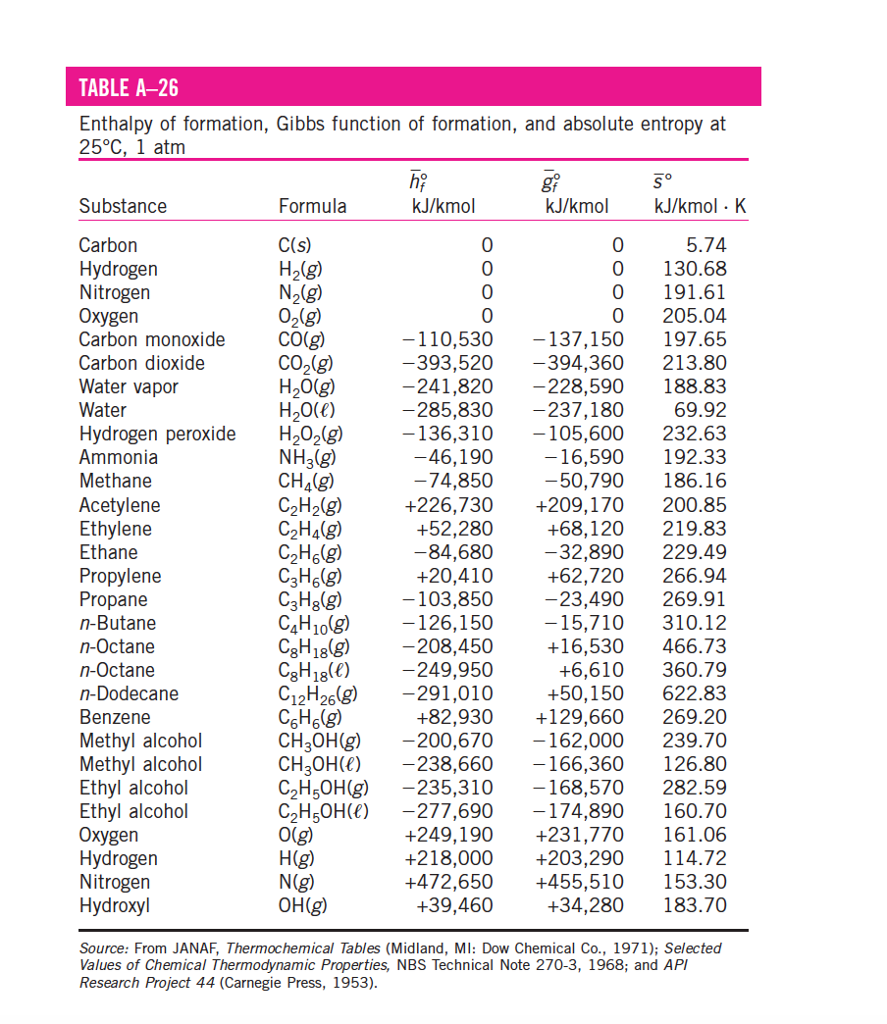
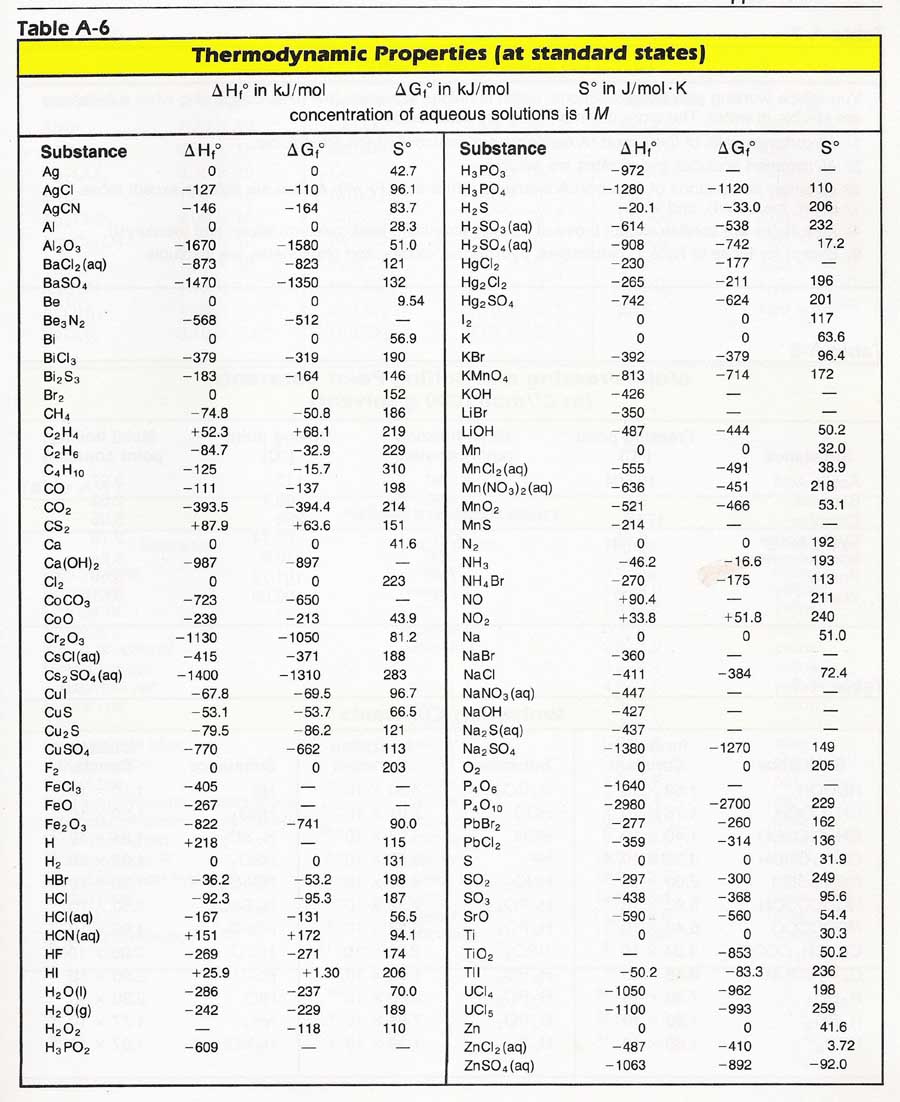
TABLE A286 Enthalpy of formation, Gibbs function of
Since enthalpy is a state function, a change in enthalpy does not depend on the pathway between two states. Hess's law: In going from a particular set of reactants to a particular set of products, the change in enthalpy is the same whether the reaction takes place in one step or in a series of steps.
Spontaneous and Nonspontaneous Reactions — Overview Expii
This table lists the standard enthalpies (ΔH°), the free energies (ΔG°) of formation of compounds from elements in their standard states, and the thermodynamic (third-law) entropies (S°) of compounds at 298 K. The state of the compound is specified by the following symbols: (g) - gas (l) = liquid (s) - solid (aq) = aqueous solution

How to Draw & Label Enthalpy Diagrams Video & Lesson Transcript
Miscellaneous Physiology Piping Systems Sanitary Drainage Systems Standard Organizations Statics Steam and Condensate Thermodynamics Water Systems Enthalpy-entropy diagram for water and steam.

Gibbs Free Energy Entropy, Enthalpy & Equilibrium Constant K YouTube
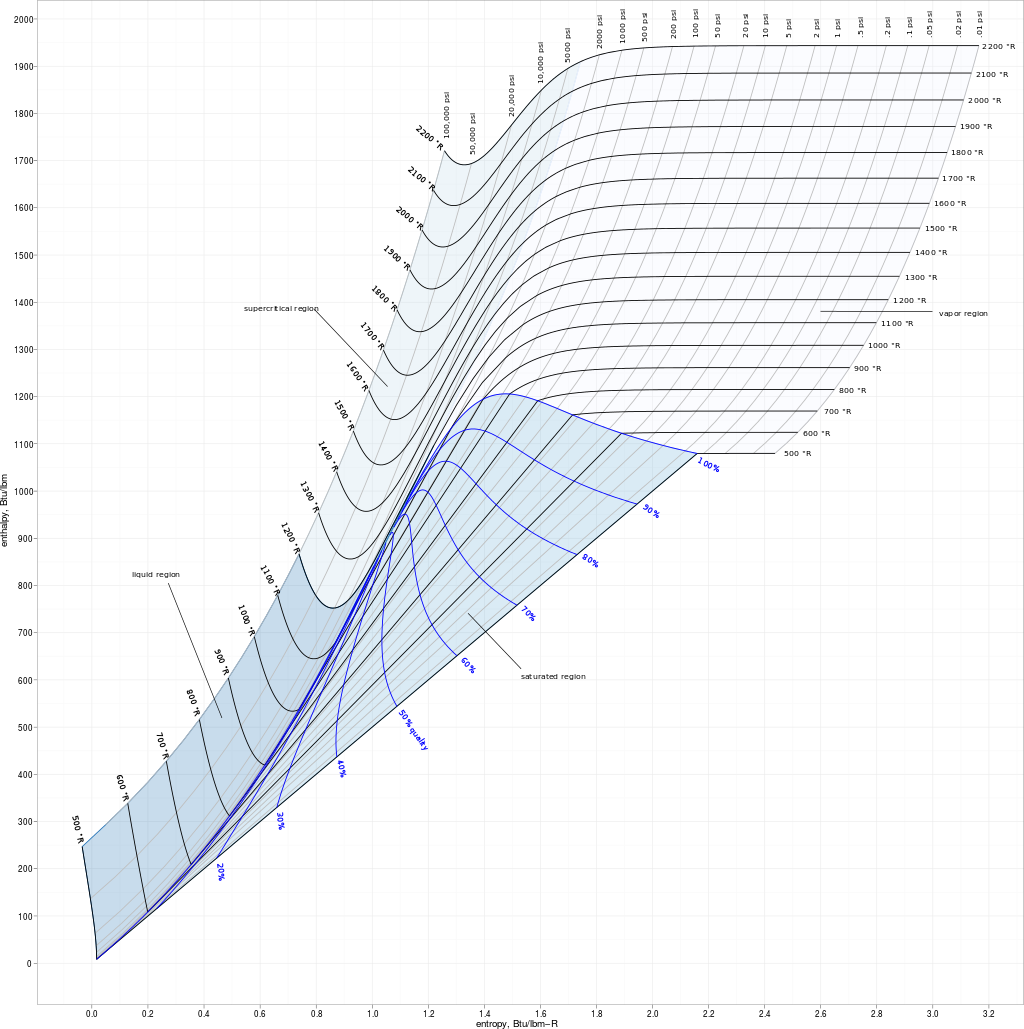
Enthalpy-entropy diagrams are presented for natural gasses of 0.6, 0.7, 0.8,0.9, and 1.0 gravity over the pressure range of 5 to 10,000 lb. per sq. in. andtemperature range of 32 degrees to 700 degrees Fahrenheit.
TEMPERATURE ENTROPY DIAGRAM FOR WATER Mechanical Engineering Professionals
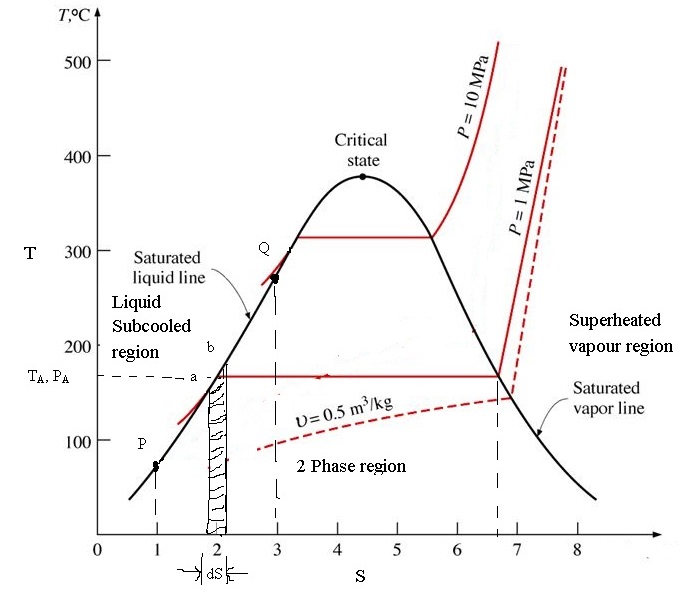
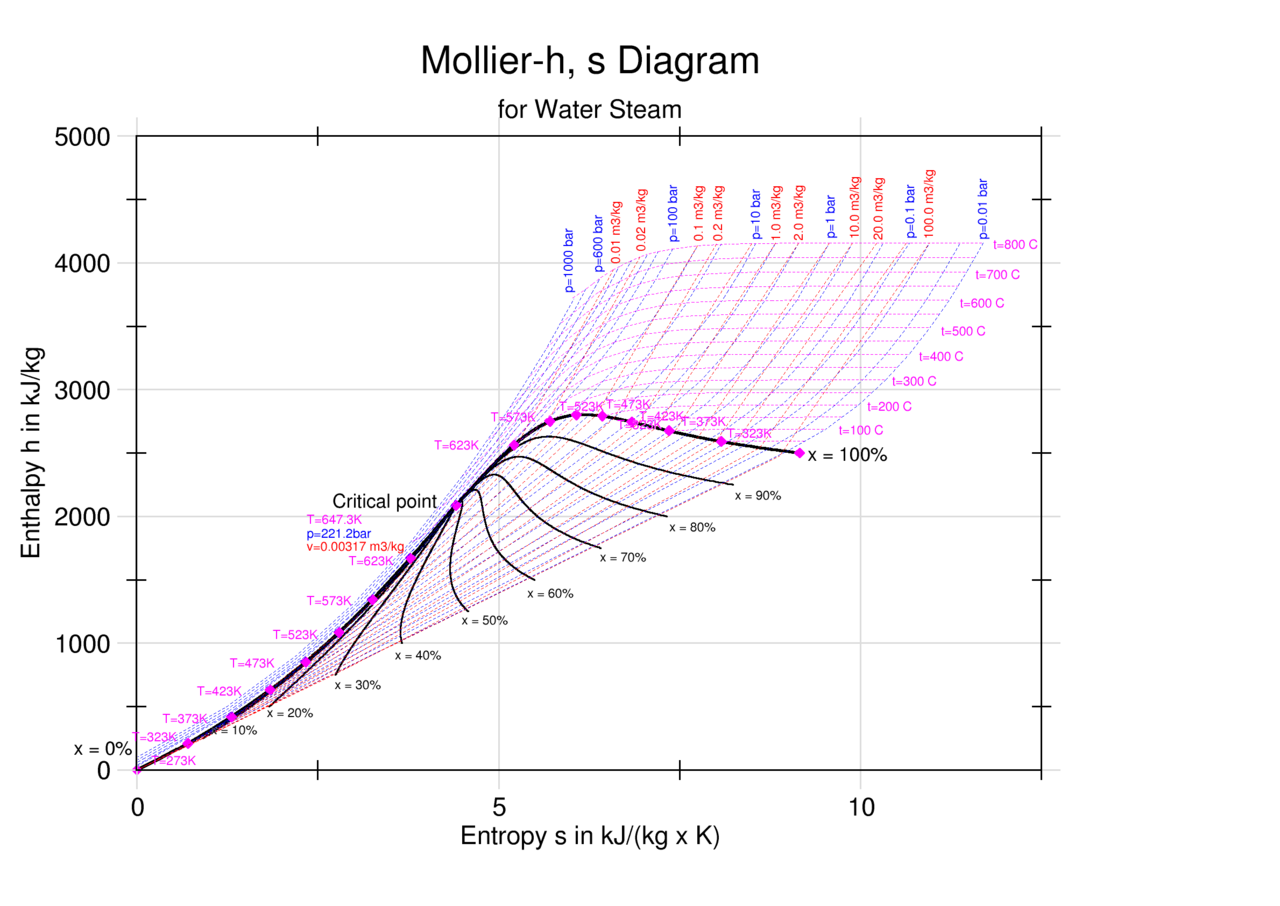
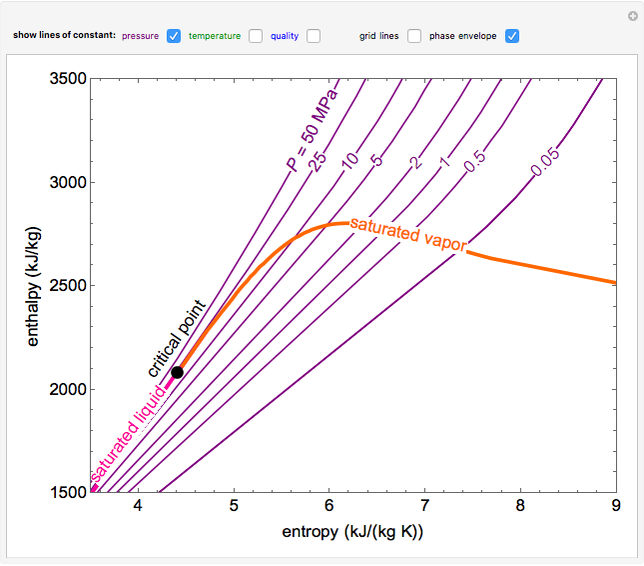
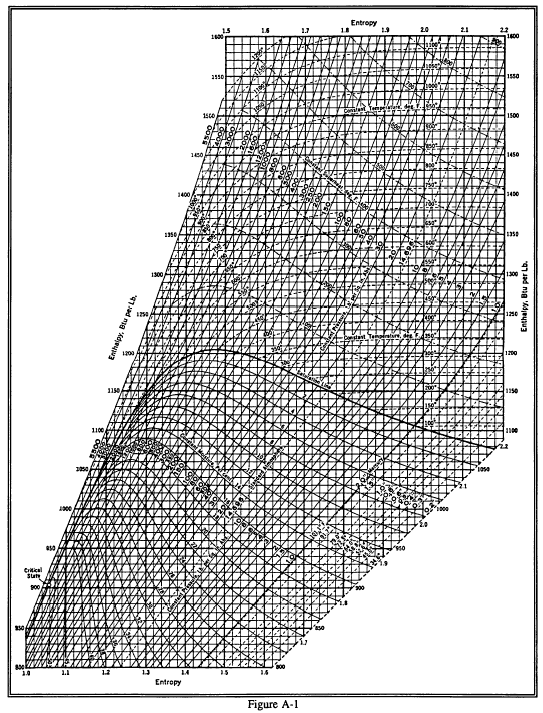
Mollier's H-S diagram (Enthalpy v Entropy) was a logical extension of the T-S diagram (Temperature v Entropy) first proposed by Gibbs, retaining the advantages of T-S diagrams but introducing several new advantages. A typical H-S Mollier diagram for a thermodynamic fluid such as steam is shown in Figure 1 . Figure 1.
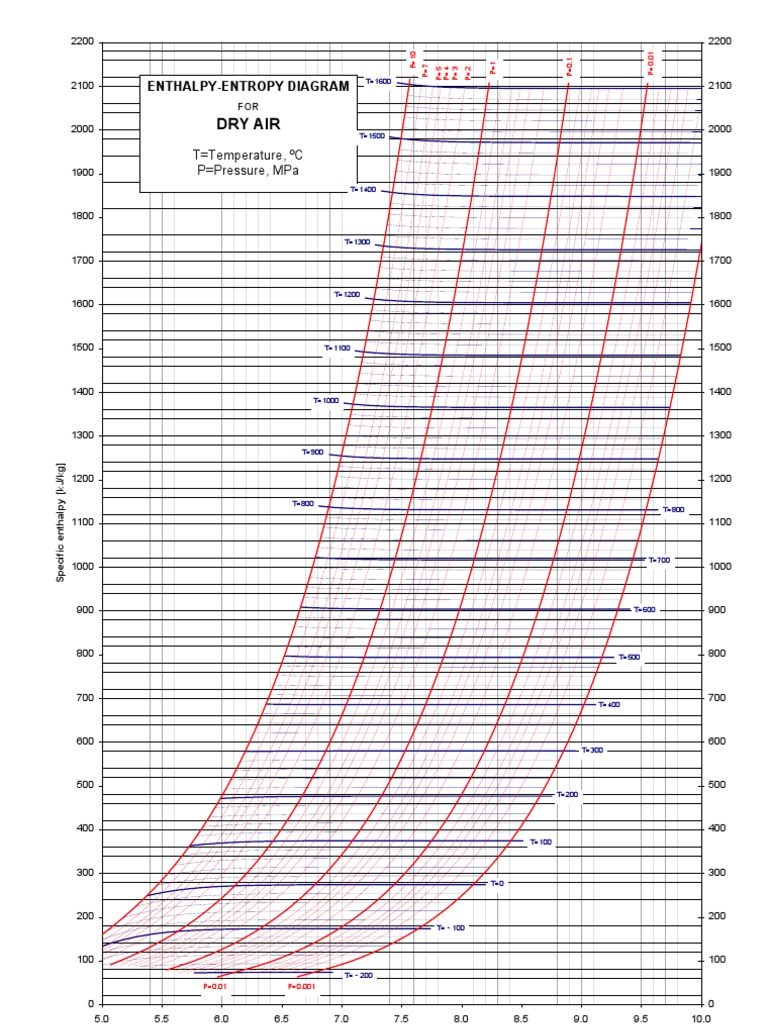
EnthalpyEntropy Diagram (Air) Enthalpy Statistical Mechanics
Of course, the main issue here is how entropy changes during a process. This can be determined by calculation from standard entropy values (\ (S^ o\)) in the same way that enthalpy changes are calculated: ∑So products − ∑So reactants = ΔSorxn (6.5.2) (6.5.2) ∑ S p r o d u c t s o − ∑ S r e a c t a n t s o = Δ S r x n o.
Enthalpy Entropy (hs) or Mollier Diagram
25°C, 1 atm Properties of some common fuels and hydrocarbons Natural logarithms of the equilibrium constant Kp Generalized enthalpy departure chart Generalized entropy departure chart Psychrometric chart at 1 atm total pressure One-dimensional isentropic compressible-flow functions for an ideal gas with k 1.4 One-dimensional normal-shock functio.
enthalpyentropydiagramforwater LearnChemE
Table of Thermodynamic Values - University of Wisconsin-Madison
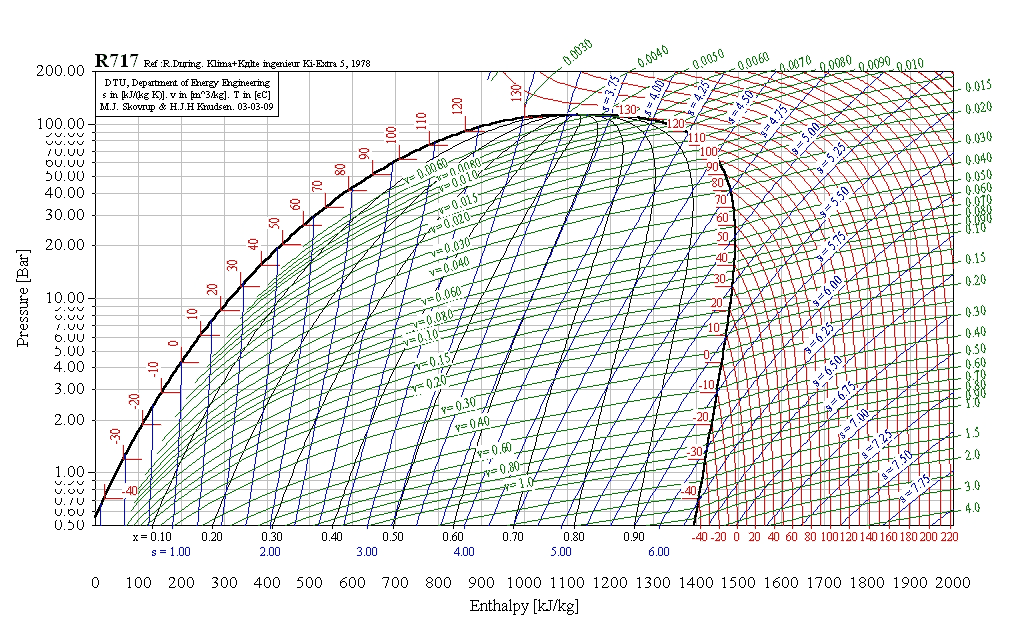
R22 Pressure Enthalpy Chart
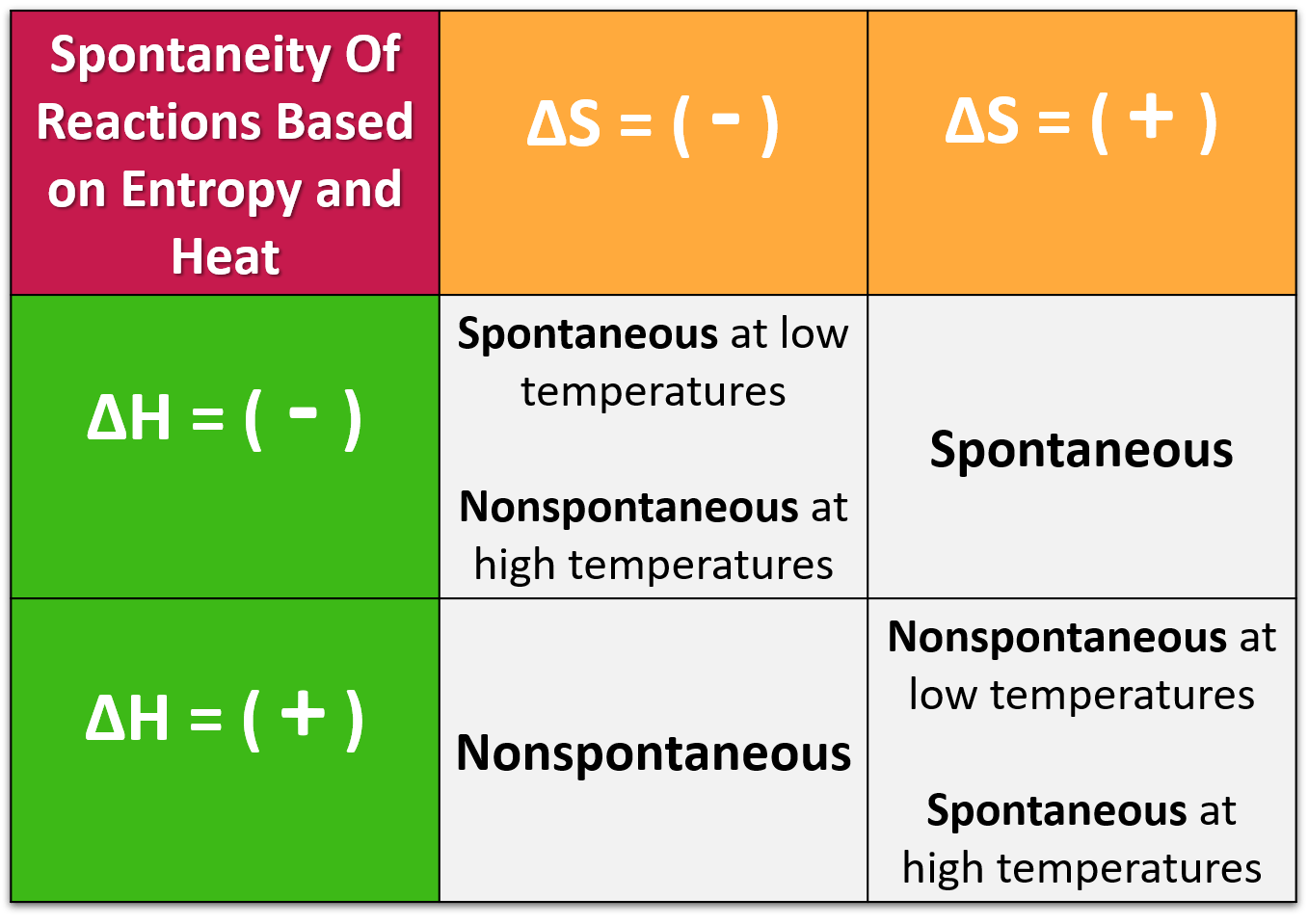
In chemical reactions involving the changes in thermodynamic quantities, a variation on this equation is often encountered: ΔGchange in free energy = ΔHchange in enthalpy − TΔS(temperature) change in entropy. Example 1.1. Calculate ∆G at 290 K for the following reaction: 2NO(g) + O 2(g) → 2NO 2(g) Given. ∆H = -120 kJ.
8 Crucial Difference between Entropy and Enthalpy with Table Core Differences
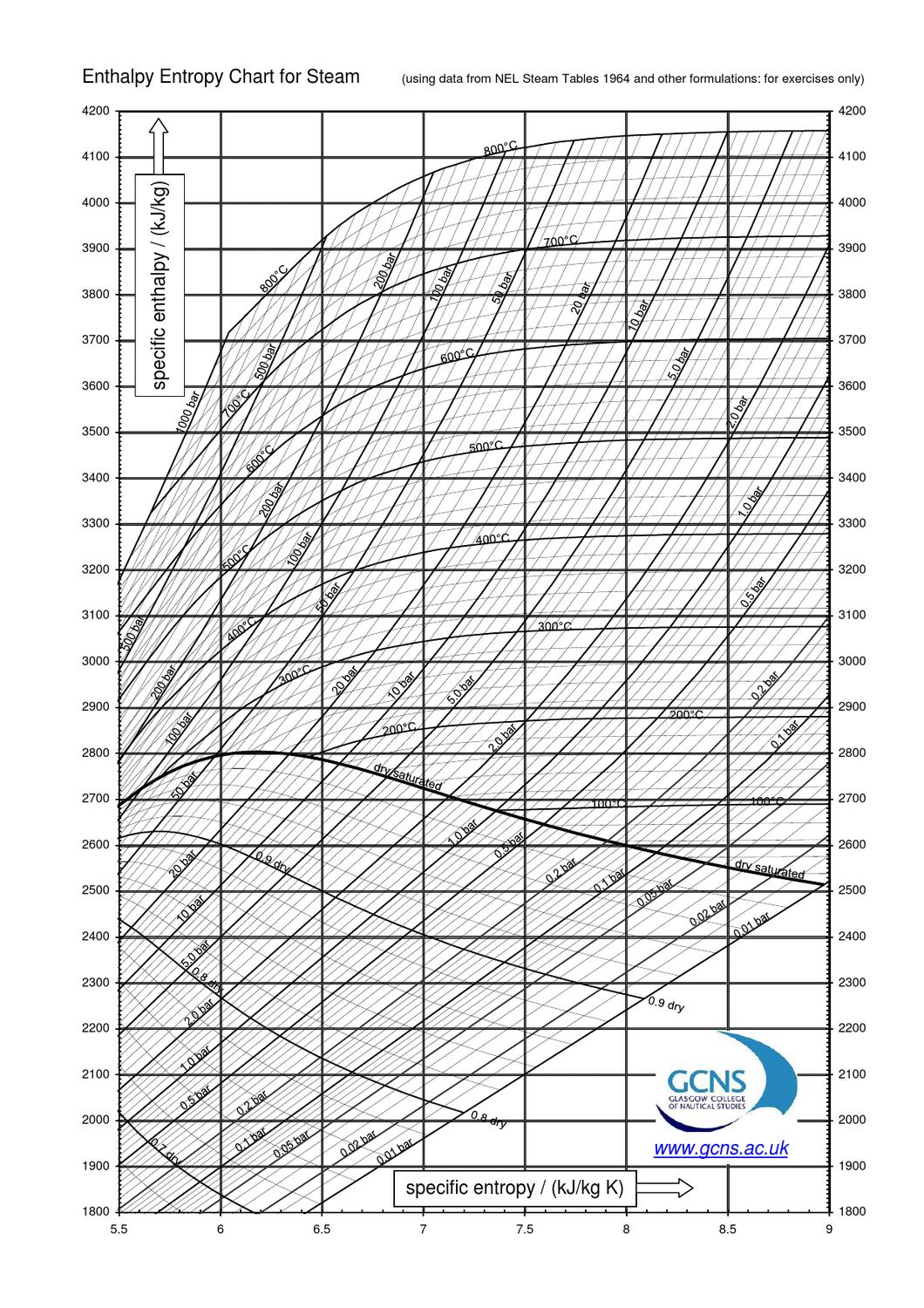
The Mollier diagram, also called the enthalpy (h) - entropy (s) chart or h-s chart, is a graphical representation of thermodynamic properties of materials. In general, it is a relationship between enthalpy (measure of the energy of a thermodynamic system), air temperature, and moisture content. Mollier Diagram Origins
Entropy Table
Details. Enthalpy and entropy are calculated using the Peng-Robinson equation of state (EOS) for a real gas and the ideal gas law for an ideal gas:. where is in kJ/mol and is in kJ/[mol K]; the superscript represents an ideal gas, the subscript refers to the reference state, and and are the enthalpy and entropy departure functions for a real gas calculated from the Peng-Robinson EOS, while.
Enthalpy Entropy (hs) or Mollier Diagram
For full table with Entropy - rotate the screen! Water - Enthalpy and Entropy vs. Temperature - SI Units [kJ/kmol] [Btu (IT)/lb] [kJ/ (kmol K)] [kJ/ (kg K)] [kWh/ (kg K)] [Btu (IT)/lb [kcal/ (kg K)] 1 Btu (IT)/lb = 0.002326 GJ/t = 2.326 kJ/kg = 0.5559 kcal/kg = 0.000646 kWh/kg
Enthalpy Entropy (hs) or Mollier Diagram Engineers Edge
34 Formula State of Matter Enthalpy (kJ/mol) Entropy (J mol/K) Gibbs Free Energy (kJ/mol) Ba 2TiO 4 (s) -2243.0424 196.648 -2133.0032 BaBr 2 (s) -757.304 146.44 -736.8024 BaBr 2 (g) -439.32 330.536 -472.792 BaBr 2•2H 2O (s) -1366.076 225.936 -1230.5144 BaCl 2 (s) -858.1384 123.67904 -810.4408 BaCl 2 (l) -832.44864 143.5112 -790.1484 BaCl 2 (g) -498.7328 325.64072 -510.69904
Enthalpy entropy diagram for a 0.65 to 0.75 specific gravity natural gas. Download Scientific
The disordered enthalpy-entropy descriptor is a mathematical formula that accelerates the computational discovery of synthesizable high-entropy ceramics, and has already guided the synthesis of.
Enthalpy Entropy Chart for Steam by Sandy Small Issuu
The Mollier diagram, shown in Figure A-1 , is a chart on which enthalpy (h) versus entropy (s) is plotted. It is sometimes known as the h-s diagram and has an entirely different shape from the T-s diagrams. The chart contains a series of constant temperature lines,a series of constant pressure lines, a series of constant moisture or quality.

Nitrogen Enthalpy, Internal Energy and Entropy vs. Temperature
An enthalpy-entropy chart, also known as the H-S chart or Mollier diagram, plots the total heat against entropy, [1] describing the enthalpy of a thermodynamic system. [2] A typical chart covers a pressure range of .01-1000 bar, and temperatures up to 800 degrees Celsius. [3]